Trusted by over 10,000 Canadians
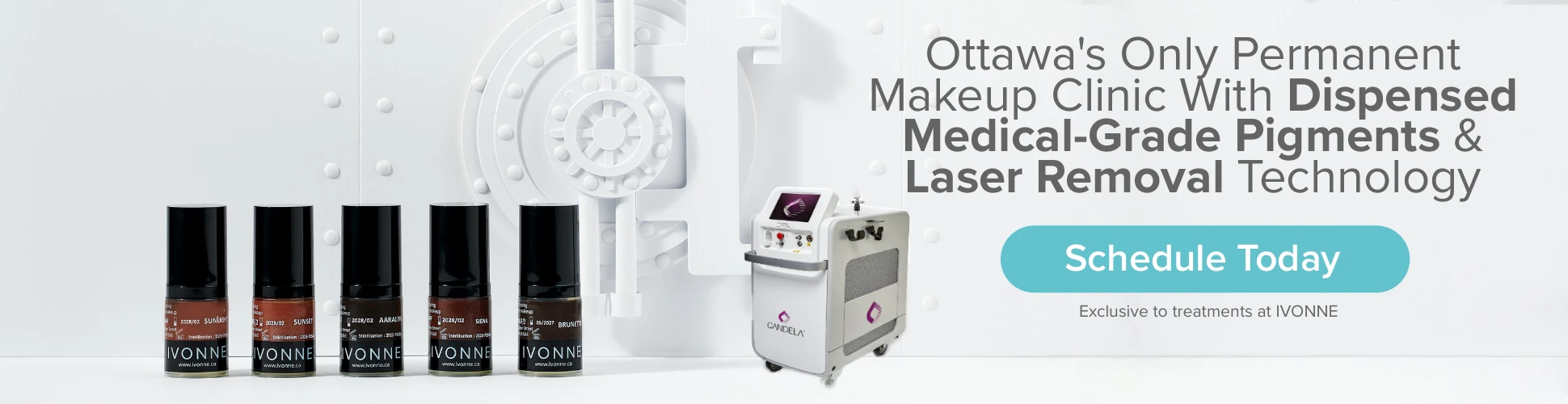
Ottawa's Specialized Permanent Makeup, Laser & Skincare Clinic
A personalized boutique experience.


Treatments With Purpose
At IVONNE clients have access to our complete flywheel of skincare, permanent makeup, and laser treatments.
IVONNE believes in the power of a holistic approach to beauty, and our flywheel allows clients to integrate all three of these services for optimal results. From skincare, to cosmetic tattoo, and removal. By taking care of their skin, enhancing their features with permanent makeup, and refreshing end-of-life PMU, our clients are able to reach their cosmetic goals.

Explore the possibilities.
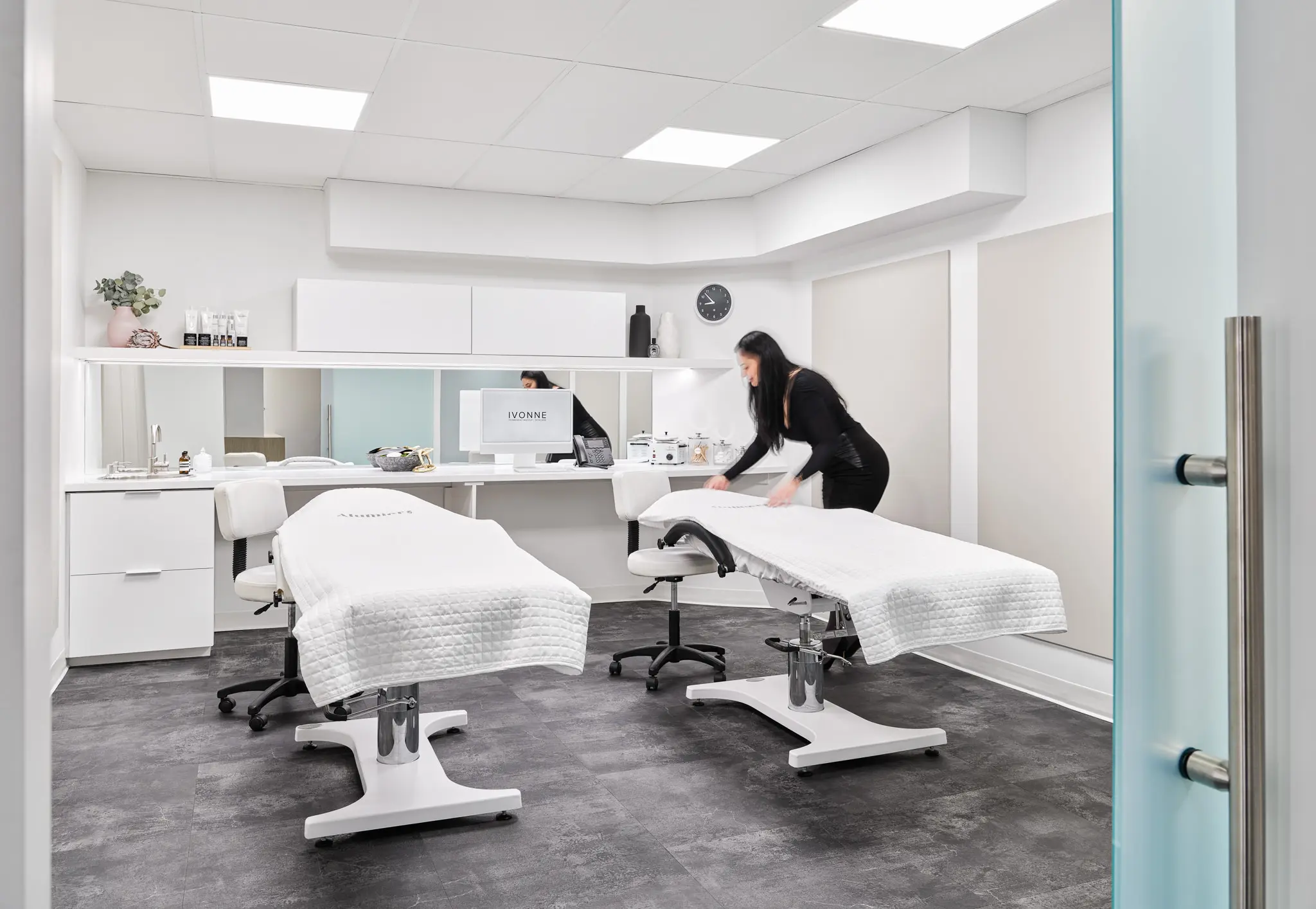
Share an experience with a friend, or colleague.
Experience private, personalized treatments.

Explore the possibilities.

Share an experience with a friend, or colleague.

Experience private, personalized treatments.
The IVONNE Experience
Each of our treatments are matched to the unique needs of every client. Experience luxurious, natural-appearing cosmetic results. Find your new favourite look in a virtual consultation. Reconnect with smooth, rejuvenated skin and take the opportunity to turn back time with clinical, science-backed treatments.
Thousands of reasons - one focus. There are as many reasons to choose IVONNE as there are skin cells in your body. From resetting tattoo ink, to new permanent brows. It's your turn to luxuriate at IVONNE.
Our Most Popular Treatments
We're Known For Our Brows
We know how important simplified self-care can be.
Our IVONNE brows are our longest-lasting brow tattoo yet. Made of biocompatible clinical-grade pigments for a more durable consistent brow colour.
Antibacterial, and anti-inflammatory. You'll enjoy longer-lasting permanent makeup with shorter, easier healing times.
Trusted by thousands of Canadians.
Image: Ivonne Sanchez, Founder
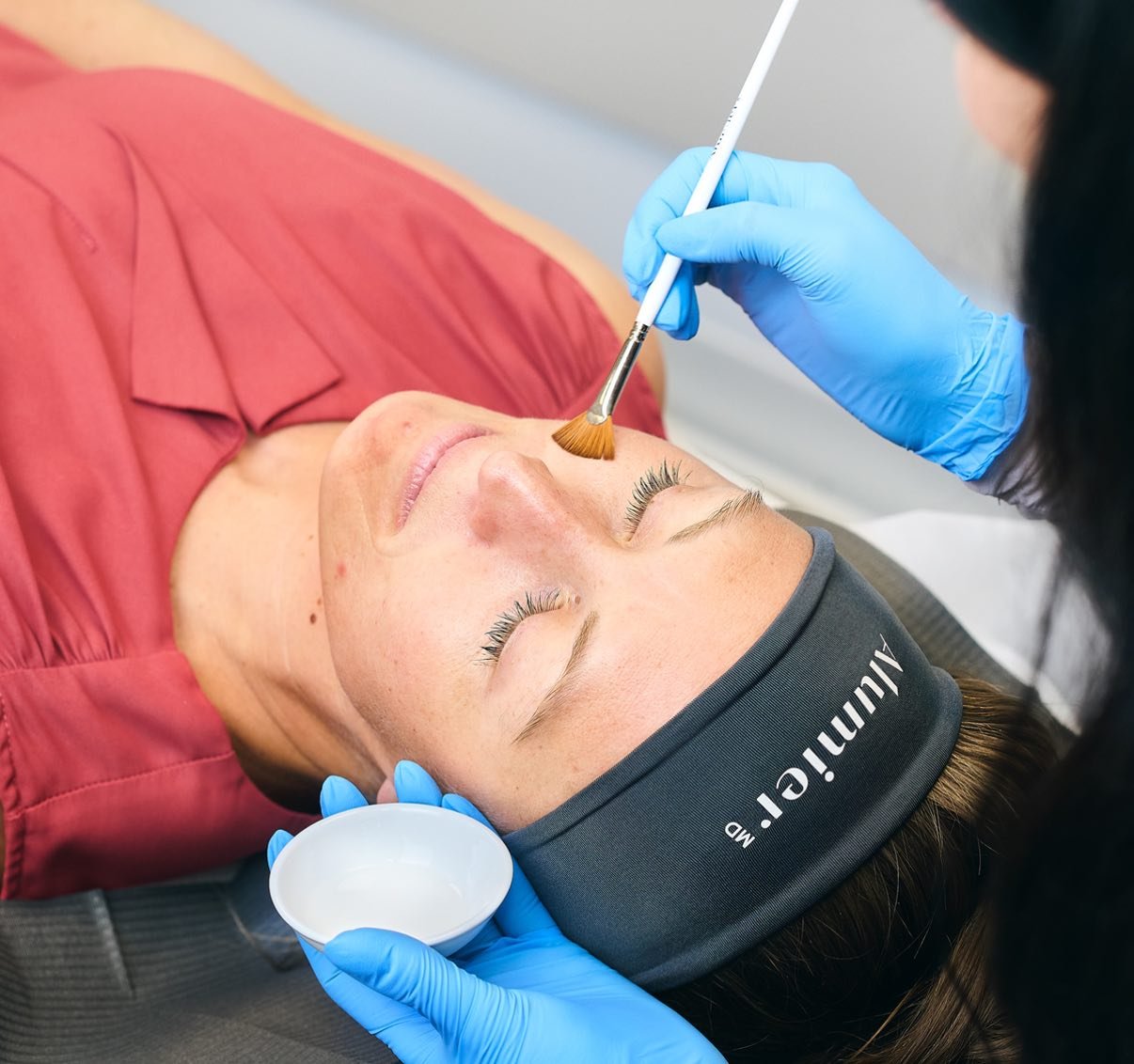
Clinical Skincare Treatments
Our Chemical Peel and Facial Treatments are backed by award-winning, science-based skincare brands like AlumierMD.
Regardless of skin type, there are facial treatments suited to everyone.
Find your new favourite skincare treatment with a personalized consultation.
Authorized Providers Of






Get your consultation today!
Get connected to a specialist and have all your questions answered. No obligation to buy.